NAVIGATION
This page is a sub-page of ObsSG, a top-level page on
the website Barbados Fringing Reefs and Seagrass Beds
(www.versicolor.ca/barbados).
Go to ObsSG for a list of other subpages in this section
of the website.
DRAFTING…
1. Earlier Lit./Manuscripts
Following are extracts related t0 successional processes in seagrass beds from a draft paper I wrote in the 1980s based on my observations at Barbados & Carriacou 1967-70.
|
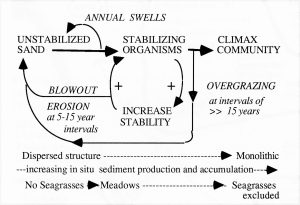
Fig. 1. Conceptual model of shallow water (<3-4m) successional processes in seagrass beds. Relationships were inferred from observations made at Barbados and Carriacou, 1968-1970. Stabilizing organisms include bottom epifauna and flora such as rhizomatous seaweeds, several species of seagrasses, and sessile fauna such as sponges, gorgonians, and corals. There is a succession of different species associated with increasing degrees of bottom stabilization (see Fig. 2). Annual swells prevent sustained establishment of early successional stages (including seagrasses). Storm events cause formation of blowouts in seagrass beds, which subsequently migrate seaward accross the beds, keeping the beds in earlier stages of succession (the most common state). In earlier and mid to later successional stages, destruction of the protective seagrass canopy by sea urchin overgrazing (associated with massive populations) can lead to large scale destruction of the beds by wave action. Very stable systems may develop monolithic structures (bound together by sponges, coralline algae) that eventually exclude seagrasses. Earlier stages of the successional sequence, cited in Patriquin, 1975, have been documented by other researchers (e.g. Williams, 1990) but no comparable schemes appear to have been elaborated for later successional stages. However an apparently unpublished, or later published manuscript – “Succession and Composition of the Thalassia Community – by Ramon Margalef and Juan Rivero from the late 1950s or early 1960s outlines a similar concept, and similar associations of species in Puerto Rico – see References Patriquin, D.G. 1975. Migration” of blowouts in seagrass beds at Barbados and Carriacou, West Indies, and its ecological and geological implications Aquatic Botany Volume 1, Pages 163-189. View PDF Williams, S.L. 1990 Experimental Studies of Caribbean Seagrass Bed Development in Ecological Monographs PDF |
2. Brigitta I. van Tussenbroek et al., 2006: Vegetation Succession*
“The sequence of succession in the seagrass beds generally involves early species that stabilize the sediments and increase the sediment nutrient content, and by doing so allow establishment of later species (Clarke and Kirkman, 1989; Williams, 1990). This suggests that succession in these beds follows the facilitation model, as is the case for many terrestrial plant communities, but uncommon for hard-bottom coastal marine areas (Connell and Slayter, 1977).
* The Biology of Thalassia: Paradigms and Recent Advances in Research Chapter 18, Brigitta I. van Tussenbroek et al., in A. W. D. Larkum et al. (eds.), 2006. Seagrasses: Biology, Ecology and Conservation, pp. 409–439. Springer. Legend: Chapter 18 The Biology of Thalassia
“In the tropical Western Atlantic, the sequence of succession is initiated, within a few months, by rhizophytic macroalgae (Fig. 2, [my Fig 3, below] Patriquin, 1975; Zieman, 1982; Williams, 1990), most commonly species of Halimeda and Penicillus, although other species of rhizophytic green algae (Table 4) can also be common colonizers.These algae supply limited amounts of nutrients to the sediment (Williams, 1990) but have minimum sediment binding capability (Zieman, 1982).”
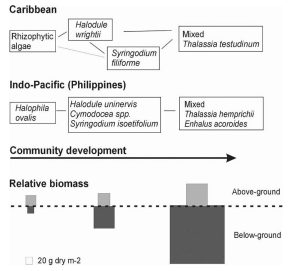 Fig 3. Idealized schemes of succession leading to a Thalassia dominated community, in the Caribbean and the Indo-Pacific. From Fig 2 in The Biology of Thalassia: Paradigms and Recent Advances in Research Chapter 18, Brigitta I. van Tussenbroek et al., in A. W. D. Larkum et al. (eds.), 2006. Seagrasses: Biology, Ecology and Conservation, pp. 409–439. Springer. Legend: Chapter 18 The Biology of Thalassia Legend to Fig. 3 (Fig 2 in Brigitta I. van Tussenbroek et al., 2006): “Idealized schemes of succession leading to a Thalassia dominated community, in the Caribbean (modified from Zieman, 1982*),and the Indo-Pacific (after a Philippine community described by Rollon et al., 1998). Later stages in succession usually contain specimen of the earlier stages. The relative biomass diagram only approximates above- and belowground biomass proportions, modified after Zieman (1982) with information from UNESCO (1998) and Duarte and Chiscano (1999) for the Caribbean and Indo-Pacific species, respectively.” * Zieman, J .C . 1982 . The ecology of the seagrasses of south Florida : a communityprofile . U.S. Fish and Wildlife Services, Office of Biological Services, Washington, D .C . FWS/QBS-82/25 . 158 pp . Two related figs from Zieman 1982: |
“In the Western Atlantic, the pioneer seagrass species is Halodule wrightii, which either colonizes from seed or through rapid vegetative spreading of nearby plants, thereby further stabilizing the sediment surface. In some sequences of succession, Syringodium filiforme appears instead of H.wrightii (Williams, 1990), or S. filiforme colonizes after the latter species, in which case the two species grow intermixed. S. filiforme is the least constant member in the sequence of succession and is frequently absent. With time and increasing development of the community, T. testudinum colonizes the region (Zieman, 1982; Williams, 1990). Its dense leaf canopy and rhizome and root system efficiently trap and retain particles, increasing the organic matter of the sediment and fueling the sedimentary microbial cycles.
“Williams (1990) experimentally removed all vegetation from 1 m 2 plots and found
that T. testudinum, after slightly more than 1 year, reached a critical density (∼200 foliar shoots m−2 ), at which it exerted control over the sedimentary nitrogen cycle. At this critical density of T. testudinum, numbers of rhizophytic algae and S. filiforme in the experimental plots declined (Williams, 1990), but she found that T. testudinum density and ammonium production rates in the sediments were still lower in her experimental plots than in the undisturbed controls at the end of the experiment (after 52 months). She suggested that T. testudinum is an atypical climax species in that is has a relatively high nutrient demands and a high (leaf) growth rate (see also Duarte and Chisano, 1999).
“…With the progression toward a climax community, there is an increase in the belowground biomass of the community as well as the leaf portion exposed in the water column, and more nutrients are sequestered by the seagrasses (Fig. 2). The increase
in leaf area provides an increase in surface area for colonization by epiphytic algae and fauna, with the surface area of the climax community being many times that of either the pioneer seagrasses or the initial algal colonizers.
“In addition to providing a substrate, the larger leaf area also increases leaf-baffling and sediment-trapping effects. Thus, as the canopy component increases, so does newly deposited material in the sediment. The climax species T. testudinum and T. hemprichii (with also E. acoroide in the Indo-Pacific), have the highest leaf area, the highest total biomass, and by far the greatest amount of material in the sediments of any species dominant in the earlier stages of succession (Fig. 2).”
3. The patch-dynamic hypothesis
– Influence of acute and chronic disturbance on macrophyte landscape zonation**
PDF
Tewfik et al., 2007 in Marine Ecology Progress Series
ABSTRACT: Although the roles of physical disturbance and successional recovery from such disturbances in structuring natural communities are well known, recent studies have begun to uncover the potential for alternate outcomes or climax states in a number of systems. Here, we examine the distribution of tropical macrophytes at a site [Bath, Barbados] with heavy wave exposure and explore equilibrium (microhabitat) and non-equilibrium (patch-dynamic) hypotheses to explain the observed pattern. The existence of a large and distinct zone of prolific macroalgae, undescribed 30 yr earlier, situated between 2 zones with a relatively high abundance of seagrass, challenges the classic successional regime within Caribbean macrophyte beds… We propose a model of macrophyte bed development and distribution that includes the important chronic ‘stress’ category of wave energy and potential dominance by macroalgae. Increased average wave energy can impose an elevated level of stress leading to modified patterns of macrophyte distributions and changes in the deterministic endpoint of the successional sequence.
**About this paper, which I first read in 2015 as I was conducting my repeat survey at Bath, I wrote Prof Guichard: “I was impressed with the interpretations in the Tewfic paper, given the limited study. There is one misconception which is the reference to “The existence of a large and distinct zone of prolific macroalgae, undescribed 30 yr earlier“… There is reference to such an assemblage in Patriquin (1975), although brief, on page 170 (“..a region of dense rhizophytic algal (Avrainvillea rawsonii and Halimeda opuntia)…”; (there is also some description to this community in my 1971 PhD thesis, but it’s buried in the appendix). However your general assumption that Avrainvillea spp have been increasing is borne out by my more detailed surveys over time.”
Prof Guichard commented in reply: “Thanks for your email, and my apologies for missing your description of extensive algal cover at Bath…As you mentioned, it was a limited study, and we at first never meant to pursue a longer term research program in that system… We have seen major changes in the seagrass-avrainvillea cover at that site; the extensive cover we described in 2003 was almost completely gone last year, a trend that had started around 2011….”
Related: Transect Observations ’69 & ’94.
More, Post 2000
Some key references.
– Dynamics of Seagrass Stability and Change
by Duarte, C.M., Fourqurean, J.W., Krause-Jensen, D., Olesen, B. (2006).
In Seagrasses: Biology, Ecology and Conservation, edited by Anthony W.D. Larkum, Robert J. Orth and Carlos M. Duarte. 2006. Springer
Duarte et al., describe how one can predict seagrass population dynamics using shoot demography and apply the techniques to understand/predicting seagrass patch and gap dynamics. The technique rests on the determination of the “Plastochrone Interval” (Patriquin, 1973; Duarte et al., 1994).